|
|
|||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||
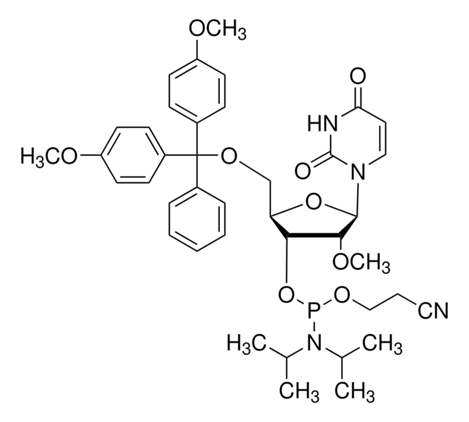 DMT-2′-O-Methyl-rU Phosphoramidite
DMT-2′-O-Methyl-rU Phosphoramidite
Catalog NO.: RNA-OMEPM-004 | CAS NO.: 110764-79-9 |
Brand: BIOCAXIS
Category
Carbohydrates, Nucleosides & Nucleotides, RNA
Phosphoramidites
Synonyms:
5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-O-methyl-uridine,
3′-[2-cyanoethyl N,N-bis(1-methylethyl)phosphoramidite], DMT-2′O-Methyl-rU
amidite
Molecular Formula
C40H49N4O9P
Molecular Weight
7601.81
General description
DMT-2′O-Methyl-rU Phosphoramidite belongs to the
class of 2′O-Methyl RNA Phosphoramidites.
Application
2′O-Methyl RNA nucleoside including DMT-2′O-Methyl-rU
Phosphoramidite can be advantageously incorporated in nucleic acid probes with
RNA or DNA for in-vivo or in-vitro applications to convey nuclease resistance.
Features and Benefits
Its key features include:
High yield of crude oligonucleotides
Compatible with DNA synthesis
Can be employed together with DNA or RNA phosphoramidites
in the same synthesis to produce mixmer oligonucleotides
Recommended deprotection conditions are 8 hours at 55 °C
using concentrated ammonia solution, or with AMA (concentrated ammonia/ 40%
aqueous methylamine I/I, v/v) for 10 minutes at 65 °C
Purification and other downstream processing of fully
modified 2′OMethyl RNA oligonucleotides are simpler than in the case of
RNA, as no special precautions are required to provide protection against
nucleolytic degradation
Synthesis of 2′O-Methyl RNA oligonucleotides is
similar to standard DNA synthesis but requires an elongated coupling time
(recommended is 6 minutes compared to 90 seconds for DNA monomers)
2′O-Methyl RNA phosphoramidites are also available
with fast deprotection chemistry
Other Notes
The unique combination of properties of 2′O-Methyl
RNA had found widespread use in the fields of:
Diagnostic probes
Aptamer and ribozyme development
Mixed 2′O-Methyl-RNA/DNA antisense molecules
Usage
Solid phase synthesis of DNA and RNA oligonucleotides in
chemical or enzymatic processes.

